

CN 11-4766/R
主办:中国科学院心理研究所
出版:科学出版社


心理科学进展 ›› 2026, Vol. 34 ›› Issue (5): 890-905.doi: 10.3724/SP.J.1042.2026.0890 cstr: 32111.14.2026.0890
收稿日期:2025-07-14
出版日期:2026-05-15
发布日期:2026-03-20
通讯作者:
刘佳丽, E-mail: ljl20240108@swu.edu.cn基金资助:
XUE Yingqi, ZHANG Yao, ZHAO Haichao, HE Qinghua, LIU Jiali( )
)
Received:2025-07-14
Online:2026-05-15
Published:2026-03-20
摘要:
路径整合是指个体在运动过程中持续地整合感觉线索以更新自身位置的空间导航能力。在人口老龄化加剧的背景下, 路径整合的行为表现及其神经特征是否可以预测早期神经退行性疾病已成为目前的研究热点。基于真实或虚拟现实环境的研究表明, 路径整合能力随正常老化至病理老化过程逐步衰退。该能力依赖于网格细胞与其他空间细胞的协作, 内嗅皮层、海马等关键节点结构和功能的衰退是导致老化过程中路径整合能力衰退的神经特征。本文旨在深入理解路径整合在老化过程中的行为衰退差异与特异性神经机制, 进而为开发老化评估诊断系统和靶向干预提供坚实的理论支撑。
中图分类号:
薛莹琦, 张瑶, 赵海潮, 何清华, 刘佳丽. (2026). 路径整合能力老化及其神经机制. 心理科学进展 , 34(5), 890-905.
XUE Yingqi, ZHANG Yao, ZHAO Haichao, HE Qinghua, LIU Jiali. (2026). Aging of path integration ability and its neural mechanisms. Advances in Psychological Science, 34(5), 890-905.

图1 PI定义及实验范式图。(A) PI示例, 沙蚁离巢觅食时沿曲折路径行进, 找到食物后能够直线返回巢穴。该过程依赖于对其自身运动信息的实时整合, 以持续计算与巢穴的相对位置关系。(B) 三角形完成范式示意图, 被试被引导依次经过路径的两个路段后, 需自主返回起点以完成三角形闭合。返回点与真实起点之间的欧式距离d为距离误差, 实际方向与目标方向之间的绝对角度差θ为角度误差。(C) 桌面式VR路径整合任务示意图, 被试在桌面VR环境中通过操纵杆移动自身位置以完成任务, 该任务排除了行走所带来的身体线索, 仅包含视觉线索。(D) 沉浸式VR路径整合任务示意图, 被试佩戴VR头显, 在VR环境中通过真实行走完成三角形任务, 该过程依赖身体和光流线索。
| 实验任务 | 具体设置 | 研究对象 | 行为表现 | 神经机制 | 参考文献 |
|---|---|---|---|---|---|
| 三角形完成任务:沿着三角形路径到达2个目标点, 再返回起始位置。 | VR头显呈现虚拟环境, 被试通过真实行走来实现运动。任务阶段分为三种条件: 1)环境与练习阶段一致(同时具备远端线索和表面纹理); 2)移除远端线索(破坏环境线索); 3)移除表面纹理(破坏视觉流) | AD风险人群(中年人) | 在移除远端线索时, AD高风险组的距离误差和角度误差显著更高。角度误差过大造成了最终位置的偏离。 | 内嗅皮层(EC)、海马亚区等体积在高低风险组间无差异, 后内侧EC网格表征信号减弱与PI损伤相关, 头朝向编码与PI损伤正相关。 | (Newton et al., |
| MCI患者、健康老年人 | MCI组的距离误差显著大于健康对照组。MCI+(脑脊液标志物为阳性)患者组的距离误差显著大于MCI-患者组和健康对照组。 | MCI组EC、后内侧EC、海马体积小于对照组, MCI+组后内侧EC体积较MCI-组减少但未达校正显著性。在所有被试中, 距离误差与EC体积和后内侧EC体积呈显著负相关, 但与海马和后扣带皮层体积无显著关联。 | (Howett et al., | ||
| 基于真实圆形空间构建了实验环境, 被试首先需预览目标三角形路径以熟悉其空间布局, 随后在视觉遮蔽条件下, 通过真实行走完成该路径。 | 健康老年人、MCI患者、AD患者 | 与健康老年人相比, MCI和AD患者的距离误差显著更高。AD组的角度误差显著高于对照组, aMCI组的角度误差与健康老年人无明显差异。 | MCI和AD患者的距离误差显著增加, 与海马体积缩小及EC变薄高度相关。 | (Mokrisova et al., | |
| 苹果游戏:被试从初始位置获取篮子, 依次到达目标点(树木), 当发现结有苹果的特殊树木后, 被试需沿最短路径返回初始位置。 | 桌面式VR呈现虚拟环境, 被试通过操纵杆实现自身位置的移动。该任务包括三个不同支持性空间线索的子任务: 1)纯粹依赖光流的子任务(pure PI, PPI); 2)具有圆形边界线索的子任务(boundary- supported PI, BPI); 3)具有地标线索的子任务(landmark- supported PI, LPI)。 该任务与三角形完成任务不同之处在于目标点数量在2~5之间变化。 | AD风险人群(18~75岁) | PPI表现显著差于BPI和LPI, LPI表现最佳。APOE4携带者在PPI中表现显著更差, 而在BPI和LPI任务中无明显差异。APOE4携带者在长返回距离时误差积累更显著。 | 在APOE4携带者中, EC体积与长返回距离下的PI表现正相关, EC和海马在返回阶段的激活与PI表现正相关, 海马在返回阶段的激活与目标接近度正相关。内侧EC的网格样表征与PPI表现相关, LPI任务中压后皮层激活显著更高。 | (Bierbrauer et al., |
| MCI患者、Aβ+个体(淀粉样蛋白阳性, 视为临床前AD)、Aβ-个体(淀粉样蛋白阴性) | 相比Aβ-组, Aβ+组在PPI中表现出更高的距离误差和角度误差, 但LPI中通过地标纠正错误, 两组表现无显著差异。 MCI患者在PPI和LPI中均存在更高的距离误差和角度误差, 且地标未能改善表现。 | 内侧颞叶tau水平与角度误差正相关, 而距离误差仅与年龄相关, 与tau或淀粉样蛋白无关。tau的影响在内侧颞叶区域特异, 额叶tau无类似关联。 海马体积缩小与LPI表现差相关, 而EC体积未显示显著关联。 | (Colmant et al., | ||
| Virtual Supermarket Task (VST) | 被试观看以第一人称视角在VR超市中移动购物推车的视频。超市中没有地标以确保被试使用自我中心策略进行导航。一旦视频停止, 被试者需要指出起点的方向和位置, 接着在地图上指出终点位置和行进方向。 | AD风险人群 | APOE4基因携带组在PI任务中的起点指向正确率显著更低, 表现出对边界的明显偏好(与环境中心偏好相对)。 | APOE4基因携带组的右侧EC与后扣带皮层连接呈现减弱趋势, 后扣带皮层与楔前叶连接呈现增强趋势。 | (Coughlan et al., |
| 路径估计任务:被试沿设定好的8条弯曲路径行走(路径转向顺序进行平衡), 每条路径有3个停止点, 被试在停止点需口头报告返回起点的距离以及与起点之间的角度。 | 该任务分为两种模态: 1)身体线索:佩戴眼罩无视觉输入, 通过真实行走完成任务, 仅依赖本体感觉和前庭线索。 2)光流线索:坐姿观看虚拟环境, 无需真实行走, 依赖光流信息。 | 健康老年人 | 老年组在身体线索和光流线索条件下的PI误差均显著高于年轻组 | 网格表征强度与老年人的PI误差呈显著负相关; 网格表征强度可独立预测老年组的PI误差, 其他因素(年龄、认知测试分数等)均无显著预测作用。 | (Stangl et al., |
表1 老年人、AD风险人群与MCI患者的PI受损情况以及神经机制
| 实验任务 | 具体设置 | 研究对象 | 行为表现 | 神经机制 | 参考文献 |
|---|---|---|---|---|---|
| 三角形完成任务:沿着三角形路径到达2个目标点, 再返回起始位置。 | VR头显呈现虚拟环境, 被试通过真实行走来实现运动。任务阶段分为三种条件: 1)环境与练习阶段一致(同时具备远端线索和表面纹理); 2)移除远端线索(破坏环境线索); 3)移除表面纹理(破坏视觉流) | AD风险人群(中年人) | 在移除远端线索时, AD高风险组的距离误差和角度误差显著更高。角度误差过大造成了最终位置的偏离。 | 内嗅皮层(EC)、海马亚区等体积在高低风险组间无差异, 后内侧EC网格表征信号减弱与PI损伤相关, 头朝向编码与PI损伤正相关。 | (Newton et al., |
| MCI患者、健康老年人 | MCI组的距离误差显著大于健康对照组。MCI+(脑脊液标志物为阳性)患者组的距离误差显著大于MCI-患者组和健康对照组。 | MCI组EC、后内侧EC、海马体积小于对照组, MCI+组后内侧EC体积较MCI-组减少但未达校正显著性。在所有被试中, 距离误差与EC体积和后内侧EC体积呈显著负相关, 但与海马和后扣带皮层体积无显著关联。 | (Howett et al., | ||
| 基于真实圆形空间构建了实验环境, 被试首先需预览目标三角形路径以熟悉其空间布局, 随后在视觉遮蔽条件下, 通过真实行走完成该路径。 | 健康老年人、MCI患者、AD患者 | 与健康老年人相比, MCI和AD患者的距离误差显著更高。AD组的角度误差显著高于对照组, aMCI组的角度误差与健康老年人无明显差异。 | MCI和AD患者的距离误差显著增加, 与海马体积缩小及EC变薄高度相关。 | (Mokrisova et al., | |
| 苹果游戏:被试从初始位置获取篮子, 依次到达目标点(树木), 当发现结有苹果的特殊树木后, 被试需沿最短路径返回初始位置。 | 桌面式VR呈现虚拟环境, 被试通过操纵杆实现自身位置的移动。该任务包括三个不同支持性空间线索的子任务: 1)纯粹依赖光流的子任务(pure PI, PPI); 2)具有圆形边界线索的子任务(boundary- supported PI, BPI); 3)具有地标线索的子任务(landmark- supported PI, LPI)。 该任务与三角形完成任务不同之处在于目标点数量在2~5之间变化。 | AD风险人群(18~75岁) | PPI表现显著差于BPI和LPI, LPI表现最佳。APOE4携带者在PPI中表现显著更差, 而在BPI和LPI任务中无明显差异。APOE4携带者在长返回距离时误差积累更显著。 | 在APOE4携带者中, EC体积与长返回距离下的PI表现正相关, EC和海马在返回阶段的激活与PI表现正相关, 海马在返回阶段的激活与目标接近度正相关。内侧EC的网格样表征与PPI表现相关, LPI任务中压后皮层激活显著更高。 | (Bierbrauer et al., |
| MCI患者、Aβ+个体(淀粉样蛋白阳性, 视为临床前AD)、Aβ-个体(淀粉样蛋白阴性) | 相比Aβ-组, Aβ+组在PPI中表现出更高的距离误差和角度误差, 但LPI中通过地标纠正错误, 两组表现无显著差异。 MCI患者在PPI和LPI中均存在更高的距离误差和角度误差, 且地标未能改善表现。 | 内侧颞叶tau水平与角度误差正相关, 而距离误差仅与年龄相关, 与tau或淀粉样蛋白无关。tau的影响在内侧颞叶区域特异, 额叶tau无类似关联。 海马体积缩小与LPI表现差相关, 而EC体积未显示显著关联。 | (Colmant et al., | ||
| Virtual Supermarket Task (VST) | 被试观看以第一人称视角在VR超市中移动购物推车的视频。超市中没有地标以确保被试使用自我中心策略进行导航。一旦视频停止, 被试者需要指出起点的方向和位置, 接着在地图上指出终点位置和行进方向。 | AD风险人群 | APOE4基因携带组在PI任务中的起点指向正确率显著更低, 表现出对边界的明显偏好(与环境中心偏好相对)。 | APOE4基因携带组的右侧EC与后扣带皮层连接呈现减弱趋势, 后扣带皮层与楔前叶连接呈现增强趋势。 | (Coughlan et al., |
| 路径估计任务:被试沿设定好的8条弯曲路径行走(路径转向顺序进行平衡), 每条路径有3个停止点, 被试在停止点需口头报告返回起点的距离以及与起点之间的角度。 | 该任务分为两种模态: 1)身体线索:佩戴眼罩无视觉输入, 通过真实行走完成任务, 仅依赖本体感觉和前庭线索。 2)光流线索:坐姿观看虚拟环境, 无需真实行走, 依赖光流信息。 | 健康老年人 | 老年组在身体线索和光流线索条件下的PI误差均显著高于年轻组 | 网格表征强度与老年人的PI误差呈显著负相关; 网格表征强度可独立预测老年组的PI误差, 其他因素(年龄、认知测试分数等)均无显著预测作用。 | (Stangl et al., |
| 细胞名称 | 主要分布位置 | 核心功能 | 特征与作用 |
|---|---|---|---|
| 网格细胞 | 内侧EC | 生成空间坐标 | 空间度量基础:周期性六边形放电模式(Fyhn et al., 情境依赖性:稳定性受环境中线索类型的影响(Lv et al., |
| 位置细胞 | 海马 | 位置编码 | 核心定位功能:选择性放电标记动物所处的位置(Hafting et al., 误差校正机制:被外部环境线索动态调节, 补偿和减少PI的累计误差(Sheffield & Dombeck, |
| 头朝向细胞 | 海马后下托、丘脑、被盖背侧核等区域 | 方向编码 | 方向编码功能:在特定方向表现出最大放电率(Taube, 稳定网格编码:头朝向信息调节网格细胞的放电模式(Winter et al., |
| 速度细胞 | 内侧EC | 速度编码 | 速度编码功能:编码动物移动的瞬时运动速度(Góis & Tort, 协同网格细胞:为距离计算提供速度输入(Dannenberg et al., |
表2 路径整合相关的空间编码细胞
| 细胞名称 | 主要分布位置 | 核心功能 | 特征与作用 |
|---|---|---|---|
| 网格细胞 | 内侧EC | 生成空间坐标 | 空间度量基础:周期性六边形放电模式(Fyhn et al., 情境依赖性:稳定性受环境中线索类型的影响(Lv et al., |
| 位置细胞 | 海马 | 位置编码 | 核心定位功能:选择性放电标记动物所处的位置(Hafting et al., 误差校正机制:被外部环境线索动态调节, 补偿和减少PI的累计误差(Sheffield & Dombeck, |
| 头朝向细胞 | 海马后下托、丘脑、被盖背侧核等区域 | 方向编码 | 方向编码功能:在特定方向表现出最大放电率(Taube, 稳定网格编码:头朝向信息调节网格细胞的放电模式(Winter et al., |
| 速度细胞 | 内侧EC | 速度编码 | 速度编码功能:编码动物移动的瞬时运动速度(Góis & Tort, 协同网格细胞:为距离计算提供速度输入(Dannenberg et al., |
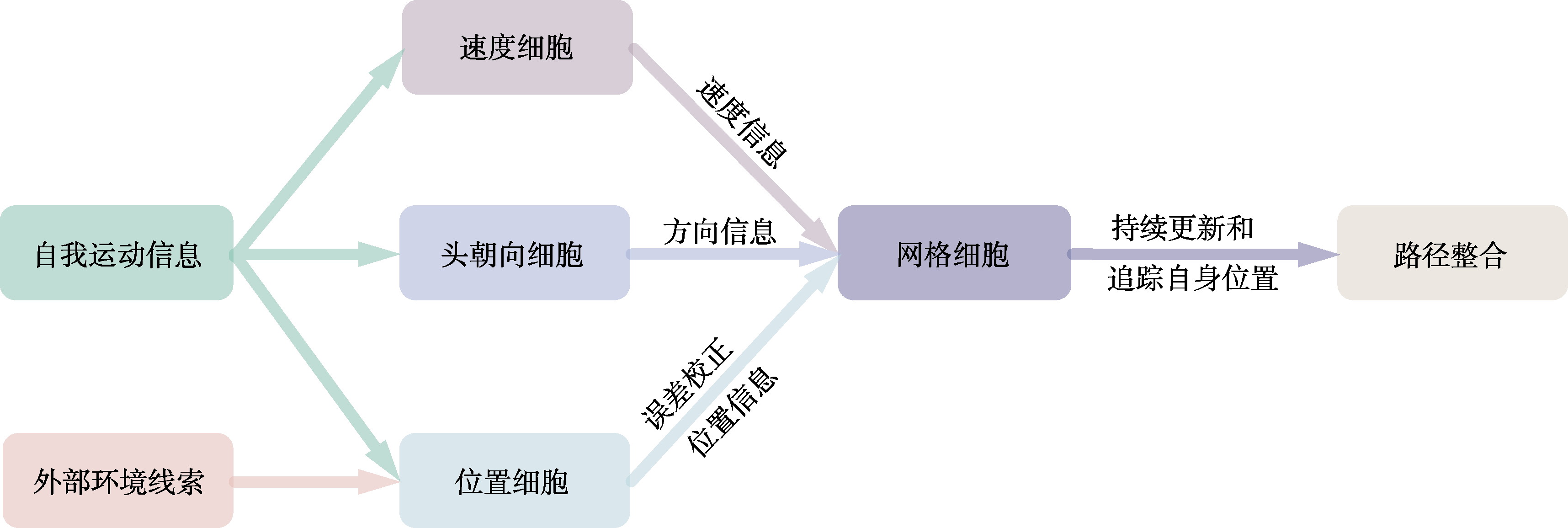
图2 网格细胞与空间编码相关细胞协作示意图。在PI过程中, 速度细胞与头朝向细胞分别对自我运动信息进行初步处理, 并将其输出的速度与方向信息传递至网格细胞。位置细胞则整合了外部环境线索与自我运动信息:在缺乏可靠外部线索时, 位置细胞主要依赖自我运动信息以维持位置编码; 而当外部线索可用时, 则可借助其对PI过程中产生的累积误差进行校正。网格细胞正是通过与上述各类空间编码细胞的协同作用, 持续更新和追踪自身位置, 从而完成PI。
| [1] |
孔祥祯, 张凤翔, 蒲艺. (2023). 空间导航的脑网络基础和调控机制. 心理科学进展, 31(3), 330-337. https://doi.org/10.3724/SP.J.1042.2023.00330
doi: 10.3724/SP.J.1042.2023.00330 URL |
| [2] |
张凤翔, 陈美璇, 蒲艺, 孔祥祯. (2023). 空间导航能力个体差异的多层次形成机制. 心理科学进展, 31(9), 1642-1664. https://doi.org/10.3724/SP.J.1042.2023.01642
doi: 10.3724/SP.J.1042.2023.01642 URL |
| [3] |
张家鑫, 海拉干, 李会杰. (2019). 空间导航的测量及其在认知老化中的应用. 心理科学进展, 27(12), 2019-2033. https://doi.org/10.3724/SP.J.1042.2019.02019
doi: 10.3724/SP.J.1042.2019.02019 URL |
| [4] |
赵辰豪, 吴德伟, 韩昆, 朱浩男, 代传金. (2021). 基于多尺度网格细胞的路径整合模型. 系统工程与电子技术, 43(10), 2961-2967. https://doi.org/10.12305/j.issn.1001-506X.2021.10.31
doi: 10.12305/j.issn.1001-506X.2021.10.31 URL |
| [5] |
Aisen P. S., Jimenez-Maggiora G. A., Rafii M. S., Walter S., & Raman R. (2022). Early-stage Alzheimer disease: Getting trial-ready. Nature Reviews Neurology, 18(7), 389-399. https://doi.org/10.1038/s41582-022-00645-6
doi: 10.1038/s41582-022-00645-6 URL pmid: 35379951 |
| [6] |
Akan O., Bierbrauer A., Axmacher N., & Wolf O. T. (2023). Acute stress impairs visual path integration. Neurobiology of Stress, 26, 100561. https://doi.org/10.1016/j.ynstr.2023.100561
doi: 10.1016/j.ynstr.2023.100561 URL |
| [7] |
Akan O., Bierbrauer A., Kunz L., Gajewski P. D., Getzmann S., Hengstler J. G., … Wolf O. T. (2023). Chronic stress is associated with specific path integration deficits. Behavioural Brain Research, 442, 114305. https://doi.org/10.1016/j.bbr.2023.114305
doi: 10.1016/j.bbr.2023.114305 URL |
| [8] |
Alexander A. S., & Nitz D. A. (2017). Spatially periodic activation patterns of retrosplenial cortex encode route sub-spaces and distance traveled. Current Biology, 27(11), 1551-1560. https://doi.org/10.1016/j.cub.2017.04.036
doi: S0960-9822(17)30480-3 URL pmid: 28528904 |
| [9] |
Allison S. L., Fagan A. M., Morris J. C., & Head D. (2016). Spatial navigation in preclinical Alzheimer’s disease. Journal of Alzheimer’s Disease, 52(1), 77-90. https://doi.org/10.3233/JAD-150855
doi: 10.3233/JAD-150855 URL |
| [10] | Arnold A. E. G. F., Burles F., Bray S., Levy R. M., & Iaria G. (2014). Differential neural network configuration during human path integration. Frontiers in Human Neuroscience, 8, 263. https://doi.org/10.3389/fnhum.2014.00263 |
| [11] |
Banino A., Barry C., Uria B., Blundell C., Lillicrap T., Mirowski P., … Kumaran D. (2018). Vector-based navigation using grid-like representations in artificial agents. Nature, 557(7705), 429-433. https://doi.org/10.1038/s41586-018-0102-6
doi: 10.1038/s41586-018-0102-6 URL |
| [12] |
Bates S. L., & Wolbers T. (2014). How cognitive aging affects multisensory integration of navigational cues. Neurobiology of Aging, 35(12), 2761-2769. https://doi.org/10.1016/j.neurobiolaging.2014.04.003
doi: S0197-4580(14)00293-0 URL pmid: 24952995 |
| [13] |
Baumann O., & Mattingley J. B. (2021). Extrahippocampal contributions to spatial navigation in humans: A review of the neuroimaging evidence. Hippocampus, 31(7), 640-657. https://doi.org/10.1002/hipo.23313
doi: 10.1002/hipo.23313 URL pmid: 33595156 |
| [14] |
Beanato E., Moon H.-J., Windel F., Vassiliadis P., Wessel M. J., Popa T., … Hummel F. C. (2024). Noninvasive modulation of the hippocampal-entorhinal complex during spatial navigation in humans. Science Advances, 10(44), eado4103. https://doi.org/10.1126/sciadv.ado4103
doi: 10.1126/sciadv.ado4103 URL |
| [15] |
Bicanski A., & Burgess N. (2020). Neuronal vector coding in spatial cognition. Nature Reviews Neuroscience, 21(9), 453-470. https://doi.org/10.1038/s41583-020-0336-9
doi: 10.1038/s41583-020-0336-9 URL pmid: 32764728 |
| [16] |
Bierbrauer A., Kunz L., Gomes C. A., Luhmann M., Deuker L., Getzmann S., … Axmacher N. (2020). Unmasking selective path integration deficits in Alzheimer’s disease risk carriers. Science Advances, 6(35), eaba1394. https://doi.org/10.1126/sciadv.aba1394
doi: 10.1126/sciadv.aba1394 URL |
| [17] | Bjerknes T. L., Dagslott N. C., Moser E. I., & Moser M.-B. (2018). Path integration in place cells of developing rats. Proceedings of the National Academy of Sciences, 115(7), 1637-1646. https://doi.org/10.1073/pnas.1719054115 |
| [18] |
Burgess N., Barry C., & O’Keefe J. (2007). An oscillatory interference model of grid cell firing. Hippocampus, 17(9), 801-812. https://doi.org/10.1002/hipo.20327
doi: 10.1002/hipo.20327 URL pmid: 17598147 |
| [19] |
Castegnaro A., Ji Z., Rudzka K., Chan D., & Burgess N. (2023). Overestimation in angular path integration precedes Alzheimer’s dementia. Current Biology, 33(21), 4650-4661. https://doi.org/10.1016/j.cub.2023.09.047
doi: 10.1016/j.cub.2023.09.047 URL |
| [20] |
Chen D., Axmacher N., & Wang L. (2024). Grid codes underlie multiple cognitive maps in the human brain. Progress in Neurobiology, 233, 102569. https://doi.org/10.1016/j.pneurobio.2024.102569
doi: 10.1016/j.pneurobio.2024.102569 URL |
| [21] |
Chen D., Kunz L., Lv P., Zhang H., Zhou W., Liang S., … Wang L. (2021). Theta oscillations coordinate grid-like representations between ventromedial prefrontal and entorhinal cortex. Science Advances, 7(44), eabj0200. https://doi.org/10.1126/sciadv.abj0200
doi: 10.1126/sciadv.abj0200 URL |
| [22] |
Chen D., Kunz L., Wang W., Zhang H., Wang W.-X., Schulze-Bonhage A., … Wang L. (2018). Hexadirectional modulation of theta power in human entorhinal cortex during spatial navigation. Current Biology, 28(20), 3310-3315.e4. https://doi.org/10.1016/j.cub.2018.08.029
doi: S0960-9822(18)31113-8 URL pmid: 30318350 |
| [23] |
Chen G., King J. A., Burgess N., & O’Keefe J. (2013). How vision and movement combine in the hippocampal place code. Proceedings of the National Academy of Sciences of the United States of America, 110(1), 378-383. https://doi.org/10.1073/pnas.1215834110
doi: 10.1073/pnas.1215834110 URL pmid: 23256159 |
| [24] |
Choo I. H., Lee D. Y., Oh J. S., Lee J. S., Lee D. S., Song I. C., … Woo J. I. (2010). Posterior cingulate cortex atrophy and regional cingulum disruption in mild cognitive impairment and Alzheimer’s disease. Neurobiology of Aging, 31(5), 772-779. https://doi.org/10.1016/j.neurobiolaging.2008.06.015
doi: 10.1016/j.neurobiolaging.2008.06.015 URL pmid: 18687503 |
| [25] | Chrastil E. R., Sherrill K. R., Aselcioglu I., Hasselmo M. E., & Stern C. E. (2017). Individual differences in human path integration abilities correlate with gray matter volume in retrosplenial cortex, hippocampus, and medial prefrontal cortex. eNeuro, 4(2), ENEURO.0346-16.2017. https://doi.org/10.1523/ENEURO.0346-16.2017 |
| [26] |
Chrastil E. R., Sherrill K. R., Hasselmo M. E., & Stern C. E. (2015). There and back again: Hippocampus and retrosplenial cortex track homing distance during human path integration. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 35(46), 15442-15452. https://doi.org/10.1523/JNEUROSCI.1209-15.2015
doi: 10.1523/JNEUROSCI.1209-15.2015 URL |
| [27] | Chrastil E. R., Sherrill K. R., Hasselmo M. E., & Stern C. E. (2016). Which way and how far? Tracking of translation and rotation information for human path integration. Human Brain Mapping, 37(10), 36-55. https://doi.org/10.1002/hbm.23265 |
| [28] | Colmant L., Quenon L., Huyghe L., Ivanoiu A., Gérard T., Lhommel R., … Hanseeuw B. (2025). Rotation errors in path integration are associated with Alzheimer’s disease tau pathology: A cross-sectional study. Alzheimer’s Research & Therapy, 17(1), 34. https://doi.org/10.1186/s13195-025-01679-w |
| [29] |
Coluccia E., & Louse G. (2004). Gender differences in spatial orientation: A review. Journal of Environmental Psychology, 24(3), 329-340. https://doi.org/10.1016/j.jenvp.2004.08.006
doi: 10.1016/j.jenvp.2004.08.006 URL |
| [30] |
Coughlan G., Plumb W., Zhukovsky P., Aung M. H., & Hornberger M. (2023). Vestibular contribution to path integration deficits in “at-genetic-risk” for Alzheimer’s disease. PloS One, 18(1), e0278239. https://doi.org/10.1371/journal.pone.0278239
doi: 10.1371/journal.pone.0278239 URL |
| [31] |
Coughlan G., Zhukovsky P., Puthusseryppady V., Gillings R., Minihane A.-M., Cameron D., & Hornberger M. (2020). Functional connectivity between the entorhinal and posterior cingulate cortices underpins navigation discrepancies in at-risk Alzheimer’s disease. Neurobiology of Aging, 90, 110-118. https://doi.org/10.1016/j.neurobiolaging.2020.02.007
doi: S0197-4580(20)30034-8 URL pmid: 32171591 |
| [32] |
Coutrot A., Manley E., Goodroe S., Gahnstrom C., Filomena G., Yesiltepe D., … SPIers H. J. (2022). Entropy of city street networks linked to future spatial navigation ability. Nature, 604(7904), 104-110. https://doi.org/10.1038/s41586-022-04486-7
doi: 10.1038/s41586-022-04486-7 URL |
| [33] | Dannenberg H., Kelley C., Hoyland A., Monaghan C. K., & Hasselmo M. E. (2019). The Firing rate speed code of entorhinal speed cells differs across behaviorally relevant time scales and does not depend on medial septum inputs. The Journal of Neuroscience, 39(18), 3434-3453. https://doi.org/10.1523/JNEUROSCI.1450-18.2019 |
| [34] |
Dobric A., De Luca S. N., Seow H. J., Wang H., Brassington K., Chan S. M. H., … Vlahos R. (2022). Cigarette smoke exposure induces neurocognitive impairments and neuropathological changes in the hippocampus. Frontiers in Molecular Neuroscience, 15, 893083. https://doi.org/10.3389/fnmol.2022.893083
doi: 10.3389/fnmol.2022.893083 URL |
| [35] |
Donaldson T. N., Jennings K. T., Cherep L. A., Blankenship P. A., Blackwell A. A., Yoder R. M., & Wallace D. G. (2019). Progression and stop organization reveals conservation of movement organization during dark exploration across rats and mice. Behavioural Processes, 162, 29-38. https://doi.org/10.1016/j.beproc.2019.01.003
doi: S0376-6357(18)30400-5 URL pmid: 30684732 |
| [36] |
Dong L. L., & Fiete I. R. (2024). Grid cells in cognition: Mechanisms and function. Annual Review of Neuroscience, 47(1), 345-368. https://doi.org/10.1146/annurev-neuro-101323-112047
doi: 10.1146/annurev-neuro-101323-112047 URL pmid: 38684081 |
| [37] |
Ekstrom A. D., & Hill P. F. (2023). Spatial navigation and memory: A review of the similarities and differences relevant to brain models and age. Neuron, 111(7), 1037-1049. https://doi.org/10.1016/j.neuron.2023.03.001
doi: 10.1016/j.neuron.2023.03.001 URL pmid: 37023709 |
| [38] |
Fu H., Rodriguez G. A., Herman M., Emrani S., Nahmani E., Barrett G., … Duff K. E. (2017). Tau pathology induces excitatory neuron loss, grid cell dysfunction and spatial memory deficits reminiscent of early Alzheimer’s disease. Neuron, 93(3), 533-541.e5. https://doi.org/10.1016/j.neuron.2016.12.023
doi: 10.1016/j.neuron.2016.12.023 URL |
| [39] |
Fukawa A., Aizawa T., Yamakawa H., & Yairi I. E. (2020). Identifying core regions for path integration on medial entorhinal cortex of hippocampal formation. Brain Sciences, 10(1), 28. https://doi.org/10.3390/brainsci10010028
doi: 10.3390/brainsci10010028 URL |
| [40] |
Fyhn M., Hafting T., Treves A., Moser M.-B., & Moser E. I. (2007). Hippocampal remapping and grid realignment in entorhinal cortex. Nature, 446(7132), 190-194. https://doi.org/10.1038/nature05601
doi: 10.1038/nature05601 URL |
| [41] |
Fyhn M., Molden S., Witter M. P., Moser E. I., & Moser M.-B. (2004). Spatial representation in the entorhinal Cortex. Science, 305(5688), 1258-1264. https://doi.org/10.1126/science.1099901
doi: 10.1126/science.1099901 URL pmid: 15333832 |
| [42] |
Gil M., Ancau M., Schlesiger M. I., Neitz A., Allen K., De Marco R. J., & Monyer H. (2018). Impaired path integration in mice with disrupted grid cell firing. Nature Neuroscience, 21(1), 81-91. https://doi.org/10.1038/s41593-017-0039-3
doi: 10.1038/s41593-017-0039-3 URL pmid: 29230055 |
| [43] |
Góis Z. H. T. D., & Tort, A. B. L. (2018). Characterizing speed cells in the rat hippocampus. Cell Reports, 25(7), 1872-1884.e4. https://doi.org/10.1016/j.celrep.2018.10.054
doi: S2211-1247(18)31643-7 URL pmid: 30428354 |
| [44] |
Guo J., Huang J., & Wan X. (2019). Influence of route decision-making and experience on human path integration. Acta Psychologica, 193, 66-72. https://doi.org/10.1016/j.actpsy.2018.12.005
doi: S0001-6918(18)30354-8 URL pmid: 30594863 |
| [45] |
Hafting T., Fyhn M., Molden S., Moser M.-B., & Moser E. I. (2005). Microstructure of a spatial map in the entorhinal cortex. Nature, 436(7052), 801-806. https://doi.org/10.1038/nature03721
doi: 10.1038/nature03721 URL |
| [46] |
Hill P. F., Bermudez S., McAVan A. S., Garren J. D., Grilli M. D., Barnes C. A., & Ekstrom A. D. (2024). Age differences in spatial memory are mitigated during naturalistic navigation. Aging, Neuropsychology and Cognition, 31(6), 1106-1130. https://doi.org/10.1080/13825585.2024.2326244
doi: 10.1080/13825585.2024.2326244 URL |
| [47] |
Howett D., Castegnaro A., Krzywicka K., Hagman J., Marchment D., Henson R., … Chan D. (2019). Differentiation of mild cognitive impairment using an entorhinal cortex-based test of virtual reality navigation. Brain, 142(6), 1751-1766. https://doi.org/10.1093/brain/awz116
doi: 10.1093/brain/awz116 URL pmid: 31121601 |
| [48] |
Igarashi K. M. (2023). Entorhinal cortex dysfunction in Alzheimer’s disease. Trends in Neurosciences, 46(2), 124-136. https://doi.org/10.1016/j.tins.2022.11.006
doi: 10.1016/j.tins.2022.11.006 URL |
| [49] |
Iglói K., Doeller C. F., Paradis A.-L., Benchenane K., Berthoz A., Burgess N., & Rondi-Reig L. (2015). Interaction between hippocampus and cerebellum crus I in sequence-based but not place-based navigation. Cerebral Cortex, 25(11), 4146-4154. https://doi.org/10.1093/cercor/bhu132
doi: 10.1093/cercor/bhu132 URL |
| [50] |
Izen S. C., Chrastil E. R., & Stern C. E. (2018). Resting state connectivity between medial temporal lobe regions and intrinsic cortical networks predicts performance in a path integration task. Frontiers in Human Neuroscience, 12, 415. https://doi.org/10.3389/fnhum.2018.00415
doi: 10.3389/fnhum.2018.00415 URL |
| [51] |
Jacobs J., Weidemann C. T., Miller J. F., Solway A., Burke J. F., Wei X.-X., … Kahana M. J. (2013). Direct recordings of grid-like neuronal activity in human spatial navigation. Nature Neuroscience, 16(9), 1188-1190. https://doi.org/10.1038/nn.3466
doi: 10.1038/nn.3466 URL pmid: 23912946 |
| [52] |
Jin W., Qin H., Zhang K., & Chen X. (2020). Spatial navigation. Advances in Experimental Medicine and Biology, 1284, 63-90. https://doi.org/10.1007/978-981-15-7086-5_7
doi: 10.1007/978-981-15-7086-5_7 URL pmid: 32852741 |
| [53] |
Jones M. W., & Wilson M. A. (2005). Theta rhythms coordinate hippocampal-prefrontal interactions in a spatial memory task. PLoS Biology, 3(12), e402. https://doi.org/10.1371/journal.pbio.0030402
doi: 10.1371/journal.pbio.0030402 URL |
| [54] |
Karn T., & Cinelli M. E. (2019). The effect of galvanic vestibular stimulation on path trajectory during a path integration task. Quarterly Journal of Experimental Psychology, 72(6), 1550-1560. https://doi.org/10.1177/1747021818798824
doi: 10.1177/1747021818798824 URL |
| [55] |
Király B., Domonkos A., Jelitai M., Lopes-Dos-Santos V., Martínez-Bellver S., Kocsis B., … Hangya B. (2023). The medial septum controls hippocampal supra-theta oscillations. Nature Communications, 14(1), 6159. https://doi.org/10.1038/s41467-023-41746-0
doi: 10.1038/s41467-023-41746-0 URL |
| [56] | Koike R., Soeda Y., Kasai A., Fujioka Y., Ishigaki S., Yamanaka A., … Takashima A. (2024). Path integration deficits are associated with phosphorylated tau accumulation in the entorhinal cortex. Brain Communications, 6(1), fcad359. https://doi.org/10.1093/braincomms/fcad359 |
| [57] |
Lee P.-L., Chou K.-H., Chung C.-P., Lai T.-H., Zhou J. H., Wang P.-N., & Lin C.-P. (2020). Posterior cingulate cortex network predicts Alzheimer’s disease progression. Frontiers in Aging Neuroscience, 12, 608667. https://doi.org/10.3389/fnagi.2020.608667
doi: 10.3389/fnagi.2020.608667 URL |
| [58] |
Liu B., Tian Q., & Gu Y. (2021). Robust vestibular self-motion signals in macaque posterior cingulate region. eLife, 10, e64569. https://doi.org/10.7554/eLife.64569
doi: 10.7554/eLife.64569 URL |
| [59] |
Loaiza V. M. (2024). An overview of the hallmarks of cognitive aging. Current Opinion in Psychology, 56, 101784. https://doi.org/10.1016/j.copsyc.2023.101784
doi: 10.1016/j.copsyc.2023.101784 URL |
| [60] |
Loomis J. M., Klatzky R. L., Golledge R. G., Cicinelli J. G., Pellegrino J. W., & Fry P. A. (1993). Nonvisual navigation by blind and sighted: Assessment of path integration ability. Journal of Experimental Psychology: General, 122(1), 73-91. https://doi.org/10.1037//0096-3445.122.1.73
doi: 10.1037/0096-3445.122.1.73 URL |
| [61] |
Lv P., Chen D., Zhang H., Zhou W., Wang M., Grewe P., Axmacher N., & Wang L. (2024). Context-dependent grid-like representations of theta power in human entorhinal cortex. Neuroscience Bulletin, 40(12), 1955-1959. https://doi.org/10.1007/s12264-024-01271-6
doi: 10.1007/s12264-024-01271-6 URL pmid: 39110390 |
| [62] |
Maidenbaum S., Miller J., Stein J. M., & Jacobs J. (2018). Grid-like hexadirectional modulation of human entorhinal theta oscillations. Proceedings of the National Academy of Sciences of the United States of America, 115(42), 10798-10803. https://doi.org/10.1073/pnas.1805007115
doi: 10.1073/pnas.1805007115 URL pmid: 30282738 |
| [63] |
McCracken M. K., Shayman C. S., Fino P. C., Stefanucci J. K., & Creem-Regehr S. H. (2025). A comparison of the effects of older age on homing performance in real and virtual environments. IEEE Transactions on Visualization and Computer Graphics, 31(5), 3213-3222. https://doi.org/10.1109/TVCG.2025.3549901
doi: 10.1109/TVCG.2025.3549901 URL |
| [64] |
McGilton K. S., Rivera T. M., & Dawson P. (2003). Can we help persons with dementia find their way in a new environment? Aging & Mental Health, 7(5), 363-371. https://doi.org/10.1080/1360786031000150676
doi: 10.1080/1360786031000150676 URL |
| [65] |
McNaughton B. L., Battaglia F. P., Jensen O., Moser E. I., & Moser M.-B. (2006). Path integration and the neural basis of the “cognitive map”. Nature Reviews Neuroscience, 7(8), 663-678. https://doi.org/10.1038/nrn1932
doi: 10.1038/nrn1932 URL pmid: 16858394 |
| [66] |
Merkle T., & Wehner R. (2009). How flexible is the systematic search behaviour of desert ants? Animal Behaviour, 77(5), 1051-1056. https://doi.org/10.1016/j.anbehav.2009.01.006
doi: 10.1016/j.anbehav.2009.01.006 URL |
| [67] |
Mokrisova I., Laczo J., Andel R., Gazova I., Vyhnalek M., Nedelska Z., … Hort J. (2016). Real-space path integration is impaired in Alzheimer’s disease and mild cognitive impairment. Behavioural Brain Research, 307, 150-158. https://doi.org/10.1016/j.bbr.2016.03.052
doi: 10.1016/j.bbr.2016.03.052 URL pmid: 27038766 |
| [68] |
Moon H.-J., Gauthier B., Park H.-D., Faivre N., & Blanke O. (2022). Sense of self impacts spatial navigation and hexadirectional coding in human entorhinal cortex. Communications Biology, 5(1), 406. https://doi.org/10.1038/s42003-022-03361-5
doi: 10.1038/s42003-022-03361-5 URL |
| [69] |
Morris G., & Derdikman D. (2023). The chicken and egg problem of grid cells and place cells. Trends in Cognitive Sciences, 27(2), 125-138. https://doi.org/10.1016/j.tics.2022.11.003
doi: 10.1016/j.tics.2022.11.003 URL |
| [70] |
Moser E. I., Hafting T., Fyhn M., Molden S., & Moser M.-B. (2005). Microstructure of a spatial map in the entorhinal cortex. Nature, 436(7052), 801-806. https://doi.org/10.1038/nature03721
doi: 10.1038/nature03721 URL |
| [71] |
Muessig L., Hauser J., Wills T. J., & Cacucci F. (2015). A developmental switch in place cell accuracy coincides with grid cell maturation. Neuron, 86(5), 1167-1173. https://doi.org/10.1016/j.neuron.2015.05.011
doi: 10.1016/j.neuron.2015.05.011 URL pmid: 26050036 |
| [72] |
Neuhaus A. H., & Bajbouj M. (2012). Memory enhancement and deep-brain stimulation of the entorhinal Area. The New England Journal of Medicine, 366(20), 1945-1946. https://doi.org/10.1056/NEJMc1203204
doi: 10.1056/NEJMc1203204 URL pmid: 22591306 |
| [73] |
Newman E. L., Climer J. R., & Hasselmo M. E. (2014). Grid cell spatial tuning reduced following systemic muscarinic receptor blockade. Hippocampus, 24(6), 643-655. https://doi.org/10.1002/hipo.22253
doi: 10.1002/hipo.22253 URL pmid: 24493379 |
| [74] | Newton C., Pope M., Rua C., Henson R., Ji Z., Burgess N., … Prevent Dementia Research Programme. (2024). Entorhinal-based path integration selectively predicts midlife risk of Alzheimer’s disease. Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, 20(4), 2779-2793. https://doi.org/10.1002/alz.13733 |
| [75] |
Nordlund M., Levernier N., Trippa M., Bourboulou R., Marti G., Monasson R., … Koenig-Gambini J. (2025). An independent coding scheme for idiothetic distance in the hippocampus. Current Biology, 35(17), 4199-4214.e8. https://doi.org/10.1016/j.cub.2025.07.050
doi: 10.1016/j.cub.2025.07.050 URL |
| [76] |
Page H. J. I., Walters D., & Stringer S. M. (2018). A speed-accurate self-sustaining head direction cell path integration model without recurrent excitation. Network: Computation in Neural Systems, 29(1-4), 37-69. https://doi.org/10.1080/0954898X.2018.1559960
doi: 10.1080/0954898X.2018.1559960 URL |
| [77] |
Péruch P., Borel L., Magnan J., & Lacour M. (2005). Direction and distance deficits in path integration after unilateral vestibular loss depend on task complexity. Cognitive Brain Research, 25(3), 862-872. https://doi.org/10.1016/j.cogbrainres.2005.09.012
URL pmid: 16256321 |
| [78] | Poucet B., Sargolini F., Song E. Y., Hangya B., Fox S., & Muller R. U. (2014). Independence of landmark and self-motion-guided navigation: A different role for grid cells. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 369(1635), 20130370. https://doi.org/10.1098/rstb.2013.0370 |
| [79] |
Provencher V., Bier N., Audet T., & Gagnon L. (2008). Errorless-based techniques can improve route finding in early Alzheimer’s disease: A case study. American Journal of Alzheimer’s Disease and Other Dementias, 23(1), 47-56. https://doi.org/10.1177/1533317507307228
doi: 10.1177/1533317507307228 URL |
| [80] |
Puthusseryppady V., Cossio D., Yu S., Rezwana F., Hegarty M., Jacobs E. G., & Chrastil E. R. (2024). Less spatial exploration is associated with poorer spatial memory in midlife adults. Frontiers in Aging Neuroscience, 16, 1382801. https://doi.org/10.3389/fnagi.2024.1382801
doi: 10.3389/fnagi.2024.1382801 URL |
| [81] |
Qiu S., Miller M. I., Joshi P. S., Lee J. C., Xue C., Ni Y., … Kolachalama V. B. (2022). Multimodal deep learning for Alzheimer’s disease dementia assessment. Nature Communications, 13(1), 3404. https://doi.org/10.1038/s41467-022-31037-5
doi: 10.1038/s41467-022-31037-5 URL |
| [82] |
Rostagno A. A. (2022). Pathogenesis of Alzheimer’s disease. International Journal of Molecular Sciences, 24(1), 107. https://doi.org/10.3390/ijms24010107
doi: 10.3390/ijms24010107 URL |
| [83] |
Sapiurka M., Squire L. R., & Clark R. E. (2016). Distinct roles of hippocampus and medial prefrontal cortex in spatial and nonspatial memory. Hippocampus, 26(12), 1515-1524. https://doi.org/10.1002/hipo.22652
doi: 10.1002/hipo.22652 URL pmid: 27576311 |
| [84] |
Schimanski L. A., Lipa P., & Barnes C. A. (2013). Tracking the course of hippocampal representations during learning: When is the map required? The Journal of Neuroscience, 33(7), 3094-3106. https://doi.org/10.1523/JNEUROSCI.1348-12.2013
doi: 10.1523/JNEUROSCI.1348-12.2013 URL |
| [85] |
Schindler A., & Bartels A. (2018). Integration of visual and non-visual self-motion cues during voluntary head movements in the human brain. NeuroImage, 172, 597-607. https://doi.org/10.1016/j.neuroimage.2018.02.006
doi: S1053-8119(18)30094-6 URL pmid: 29427850 |
| [86] |
Schug M. G. (2016). Geographical cues and developmental exposure: Navigational style, wayfinding anxiety, and childhood experience in the Faroe Islands. Human Nature, 27(1), 68-81. https://doi.org/10.1007/s12110-015-9245-4
doi: 10.1007/s12110-015-9245-4 URL |
| [87] |
Segen V., Kabir M. R., Streck A., Slavik J., Glanz W., Butryn M., … Wolbers T. (2025). Path integration impairments reveal early cognitive changes in subjective cognitive decline. Science Advances, 11(36), eadw6404. https://doi.org/10.1126/sciadv.adw6404
doi: 10.1126/sciadv.adw6404 URL |
| [88] |
Segen V., Ying J., Morgan E., Brandon M., & Wolbers T. (2022). Path integration in normal aging and Alzheimer’s disease. Trends in Cognitive Sciences, 26(2), 142-158. https://doi.org/10.1016/j.tics.2021.11.001
doi: 10.1016/j.tics.2021.11.001 URL |
| [89] | Sejunaite K., Lanza C., Ganders S., Iljaitsch A., & Riepe M. W. (2017). Augmented reality: Sustaining autonomous way-finding in the community for older persons with cognitive impairment. The Journal of Frailty & Aging, 6(4), 206-211. https://doi.org/10.14283/jfa.2017.25 |
| [90] | Semenov Y. R., Bigelow R. T., Xue Q.-L., du Lac S., & Agrawal Y. (2016). Association between vestibular and cognitive function in U.S. adults: Data from the National Health and Nutrition Examination Survey. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 71(2), 243-250. https://doi.org/10.1093/gerona/glv069 |
| [91] |
Sheffield M. E., & Dombeck D. A. (2019). Dendritic mechanisms of hippocampal place field formation. Current Opinion in Neurobiology, 54, 1-11. https://doi.org/10.1016/j.conb.2018.07.004
doi: S0959-4388(18)30073-4 URL pmid: 30036841 |
| [92] | Shinder M. E., & Taube J. S. (2010). Differentiating ascending vestibular pathways to the cortex involved in spatial cognition. Journal of Vestibular Research: Equilibrium & Orientation, 20(1), 3-23. https://doi.org/10.3233/VES-2010-0344 |
| [93] |
Shine J. P., Valdés-Herrera J. P., Hegarty M., & Wolbers T. (2016). The human retrosplenial cortex and thalamus code head direction in a global reference frame. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 36(24), 6371-6381. https://doi.org/10.1523/JNEUROSCI.1268-15.2016
doi: 10.1523/JNEUROSCI.1268-15.2016 URL |
| [94] |
Shrager Y., Kirwan C. B., & Squire L. R. (2008). Neural basis of the cognitive map: Path integration does not require hippocampus or entorhinal cortex. Proceedings of the National Academy of Sciences of the United States of America, 105(33), 12034-12038. https://doi.org/10.1073/pnas.0805414105
doi: 10.1073/pnas.0805414105 URL pmid: 18687893 |
| [95] |
Sjolund L. A., Kelly J. W., & McNamara T. P. (2018). Optimal combination of environmental cues and path integration during navigation. Memory & Cognition, 46(1), 89-99. https://doi.org/10.3758/s13421-017-0747-7
doi: 10.3758/s13421-017-0747-7 URL |
| [96] |
Solstad T., Boccara C. N., Kropff E., Moser M.-B., & Moser E. I. (2008). Representation of geometric borders in the entorhinal cortex. Science, 322(5909), 1865-1868. https://doi.org/10.1126/science.1166466
doi: 10.1126/science.1166466 URL pmid: 19095945 |
| [97] |
Stangl M., Achtzehn J., Huber K., Dietrich C., Tempelmann C., & Wolbers T. (2018). Compromised grid-cell-like representations in old age as a key mechanism to explain age-related navigational deficits. Current Biology, 28(7), 1108-1115.e6. https://doi.org/10.1016/j.cub.2018.02.038
doi: S0960-9822(18)30223-9 URL pmid: 29551413 |
| [98] |
Stangl M., Kanitscheider I., Riemer M., Fiete I., & Wolbers T. (2020). Sources of path integration error in young and aging humans. Nature Communications, 11(1), 2626. https://doi.org/10.1038/s41467-020-15805-9
doi: 10.1038/s41467-020-15805-9 URL |
| [99] |
Stark S. M., & Stark, C. E. L. (2017). Age-related deficits in the mnemonic similarity task for objects and scenes. Behavioural Brain Research, 333, 109-117. https://doi.org/10.1016/j.bbr.2017.06.049
doi: S0166-4328(17)30637-X URL pmid: 28673769 |
| [100] | Sugar J., Witter M. P., van Strien N. M., & Cappaert, N. L. M. (2011). The retrosplenial cortex: Intrinsic connectivity and connections with the (para)hippocampal region in the rat. An interactive connectome. Frontiers in Neuroinformatics, 5, 7. https://doi.org/10.3389/fninf.2011.00007 |
| [101] |
Taube J. S. (2007). The head direction signal: Origins and sensory-motor integration. Annual Review of Neuroscience, 30, 181-207. https://doi.org/10.1146/annurev.neuro.29.051605.112854
URL pmid: 17341158 |
| [102] | Terstege D. J., Galea L. A. M., Epp J. R., & Alzheimer’s Disease Neuroimaging Initiative. (2024). Retrosplenial hypometabolism precedes the conversion from mild cognitive impairment to Alzheimer’s disease. Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, 20(12), 8979-8986. https://doi.org/10.1002/alz.14258 |
| [103] |
Van Cauter T., Camon J., Alvernhe A., Elduayen C., Sargolini F., & Save E. (2013). Distinct roles of medial and lateral entorhinal cortex in spatial cognition. Cerebral Cortex, 23(2), 451-459. https://doi.org/10.1093/cercor/bhs033
doi: 10.1093/cercor/bhs033 URL |
| [104] |
Weisberg S. M., & Ekstrom A. D. (2021). Hippocampal volume and navigational ability: The mapping is not to scale. Neuroscience and Biobehavioral Reviews, 126, 102-112. https://doi.org/10.1016/j.neubiorev.2021.03.012
doi: 10.1016/j.neubiorev.2021.03.012 URL pmid: 33722618 |
| [105] |
Winter S. S., Clark B. J., & Taube J. S. (2015). Disruption of the head direction cell network impairs the parahippocampal grid cell signal. Science, 347(6224), 870-874. https://doi.org/10.1126/science.1259591
doi: 10.1126/science.1259591 URL pmid: 25700518 |
| [106] |
Wolbers T., Wiener J. M., Mallot H. A., & Büchel C. (2007). Differential recruitment of the hippocampus, medial prefrontal cortex, and the human motion complex during path integration in humans. The Journal of Neuroscience, 27(35), 9408-9416. https://doi.org/10.1523/JNEUROSCI.2146-07.2007
doi: 10.1523/JNEUROSCI.2146-07.2007 URL |
| [107] |
Ying J., Keinath A. T., Lavoie R., Vigneault E., El Mestikawy S., & Brandon M. P. (2022). Disruption of the grid cell network in a mouse model of early Alzheimer’s disease. Nature Communications, 13, 886. https://doi.org/10.1038/s41467-022-28551-x
doi: 10.1038/s41467-022-28551-x URL |
| [108] |
Ying J., Reboreda A., Yoshida M., & Brandon M. P. (2023). Grid cell disruption in a mouse model of early Alzheimer’s disease reflects reduced integration of self-motion cues. Current Biology, 33(12), 2425-2437.e5. https://doi.org/10.1016/j.cub.2023.04.065
doi: 10.1016/j.cub.2023.04.065 URL |
| [109] | Yue L., Pan Y., Li W., Mao J., Hong B., Gu Z.,... Xiao S. (2025). Predicting cognitive decline: Deep-learning reveals subtle brain changes in pre-MCI stage. The Journal of Prevention of Alzheimer’s Disease, 12(5), 100079. https://doi.org/10.1016/j.tjpad.2025.100079 |
| [110] |
Zhou R., Belge T., & Wolbers T. (2023). Reaching the goal: Superior navigators in late adulthood provide a novel perspective into successful cognitive aging. Topics in Cognitive Science, 15(1), 15-45. https://doi.org/10.1111/tops.12608
doi: 10.1111/tops.v15.1 URL |
| [1] | 张军恒, 黄雷, 李奎良, 王靖, 姬鸣. 空间导航中方向感的多模态信息整合认知神经机制[J]. 心理科学进展, 2026, 34(6): 1010-1034. |
| [2] | 陈曦梅, 李为, 陈红. 基于奖赏−抑制双系统模型饮食失调的神经机制[J]. 心理科学进展, 2026, 34(4): 597-607. |
| [3] | 申玥, 辛聪, 郑远霞, 刘国雄. 儿童亲社会行为中的声誉管理及其心理机制[J]. 心理科学进展, 2026, 34(4): 726-741. |
| [4] | 郭新宇, 汤煜尧, 张丹丹. 同步TMS-EEG技术在心理学研究中的应用[J]. 心理科学进展, 2026, 34(3): 441-460. |
| [5] | 岳丽明, 刘振南, 高湘萍. 不同类型元认知反思的特异性与协同神经机制:一个整合性理论模型[J]. 心理科学进展, 2026, 34(3): 487-498. |
| [6] | 孙焕翔, 张帆, 李思嘉, 张秀玲, 蒋毅. 化繁为简:视觉集合感知的神经机制[J]. 心理科学进展, 2026, 34(2): 251-270. |
| [7] | 务凯. 东方正念的心理治疗机制与神经基础[J]. 心理科学进展, 2026, 34(2): 331-347. |
| [8] | 彭玉佳, 王愉茜, 鞠芊芊, 刘峰, 徐佳. 贝叶斯框架下社交焦虑的社会认知特性[J]. 心理科学进展, 2025, 33(8): 1267-1274. |
| [9] | 隋雪, 安禹思, 许艺楠, 李雨桐. 快速阅读的眼动特征、认知特点及神经机制[J]. 心理科学进展, 2025, 33(8): 1358-1366. |
| [10] | 何鸿, 张馨月, 石京鸿, 刘强. 转回努力训练对心智游移的影响及其机制探索[J]. 心理科学进展, 2025, 33(7): 1077-1090. |
| [11] | 余凌峰, 张婕, 明先超, 雷怡. 无意识恐惧及其神经机制[J]. 心理科学进展, 2025, 33(7): 1234-1245. |
| [12] | 薛笑然, 崔伟, 张丽. 空间导航能力性别差异的三水平元分析[J]. 心理科学进展, 2025, 33(5): 843-862. |
| [13] | 黄雷, 张军恒, 姬鸣. 视觉线索受限环境导航中认知地图的动态加工机制[J]. 心理科学进展, 2025, 33(4): 673-679. |
| [14] | 程晓荣, 仇式明, 定险峰, 范炤. 动作如何影响元认知?——基于认知模型和神经机制的探讨[J]. 心理科学进展, 2025, 33(3): 425-438. |
| [15] | 巩芳颍, 孙逸梵, 贺琴, 石可, 刘伟, 陈宁. 教学互动中师生脑间同步性及其调节因素[J]. 心理科学进展, 2025, 33(3): 452-464. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||