

CN 11-4766/R
主办:中国科学院心理研究所
出版:科学出版社


心理科学进展 ›› 2026, Vol. 34 ›› Issue (3): 499-514.doi: 10.3724/SP.J.1042.2026.0499 cstr: 32111.14.2026.0499
收稿日期:2025-05-17
出版日期:2026-03-15
发布日期:2026-01-07
通讯作者:
陈伟海, E-mail: whchen@swu.edu.cn基金资助:Received:2025-05-17
Online:2026-03-15
Published:2026-01-07
摘要:
工作记忆作为高级认知功能的核心, 依赖于前额叶−海马−内侧隔核神经环路的动态协同, 其中theta-gamma相位幅值耦合(TG-PAC)是跨脑区信息整合的关键机制。本文系统阐述了前额叶−海马−内侧隔核神经环路中theta-gamma相位幅值耦合在工作记忆中的核心调控作用。研究表明, 前额叶通过持续性神经活动维持工作记忆中的信息表征, 其theta振荡(4~8 Hz)通过相位调制gamma活动(30~80 Hz)形成认知控制的神经时间窗。海马作为空间信息处理的枢纽, 通过theta-gamma嵌套编码实现空间导航与工作记忆绑定, 其局部gamma振荡与theta振荡的耦合强度可预测记忆容量与行为表现。前额叶theta相位与海马gamma幅值的跨脑区耦合, 构成了认知控制与记忆存储的动态交互界面, 确保工作记忆任务的精准执行。内侧隔核作为关键中继节点, 其胆碱能、GABA能神经元通过调控海马theta振荡, 影响海马theta-gamma相位幅值耦合的强度与时空特性, 进而调节工作记忆效能。此外, TG-PAC异常与精神分裂症、阿尔茨海默病等认知功能障碍密切相关, 提示其作为潜在生物标志物和神经调控靶点的临床价值。本文创新性整合前额叶−海马−内侧隔核三节点环路之间的theta-gamma相位幅值耦合, 并展望未来研究需结合多模态成像、细胞特异性调控与计算建模, 以推动基于神经振荡耦合的认知障碍干预新策略。
中图分类号:
张秋霞, 陈伟海. (2026). 前额叶−海马−内侧隔核环路的theta-gamma相位幅值耦合: 跨脑区协同与工作记忆调控机制. 心理科学进展 , 34(3), 499-514.
ZHANG Qiuxia, CHEN Weihai. (2026). Theta-gamma phase-amplitude coupling in the prefrontal-hippocampal- medial septal circuit: Mechanisms of cross-regional coordination and working memory regulation. Advances in Psychological Science, 34(3), 499-514.
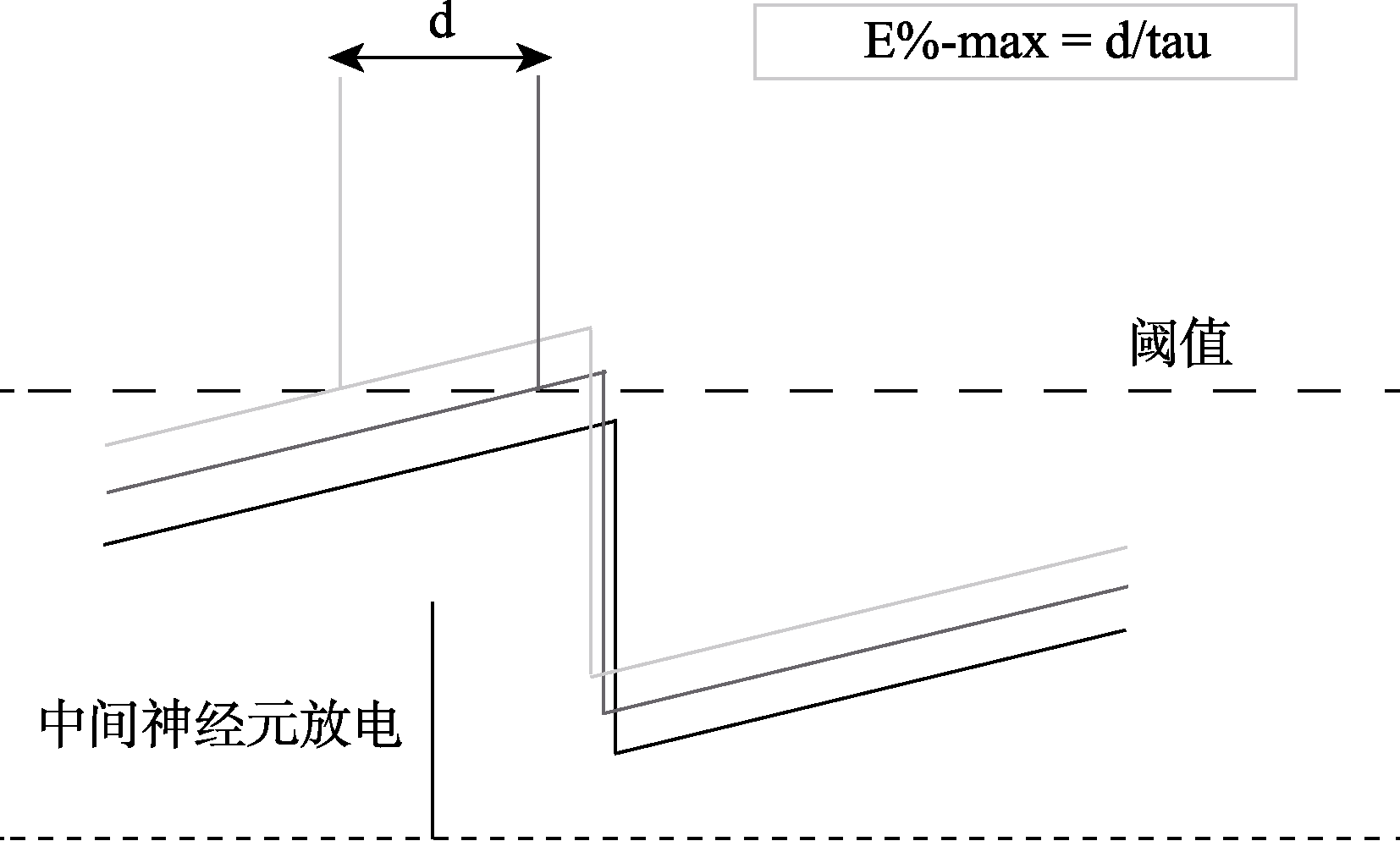
图2 gamma振荡周期中的全局性抑制如何决定哪些神经元放电。图片中三种不同灰度的直线分别代表了一个gamma周期内兴奋性由强到弱的三种神经元, 浅灰代表最兴奋的神经元, 中灰代表中等兴奋的神经元, 黑色的则代表低等兴奋神经元。在一个gamma周期中, 伴随着中间神经元对周围神经元抑制的衰减(由抑制性突触后电位 的时间常数tau决定), 最兴奋的神经元最先放电, 一旦某个神经元放电, 它会通过快速抑制性中间神经元触发全局反馈抑制, 阻止其他神经元继续放电, 然而, 由于反馈抑制的传递存在延迟(d), 在这个延迟期窗口内, 兴奋性略低于最兴奋神经元的其他神经元(中等强度的神经元)也有机会放电。
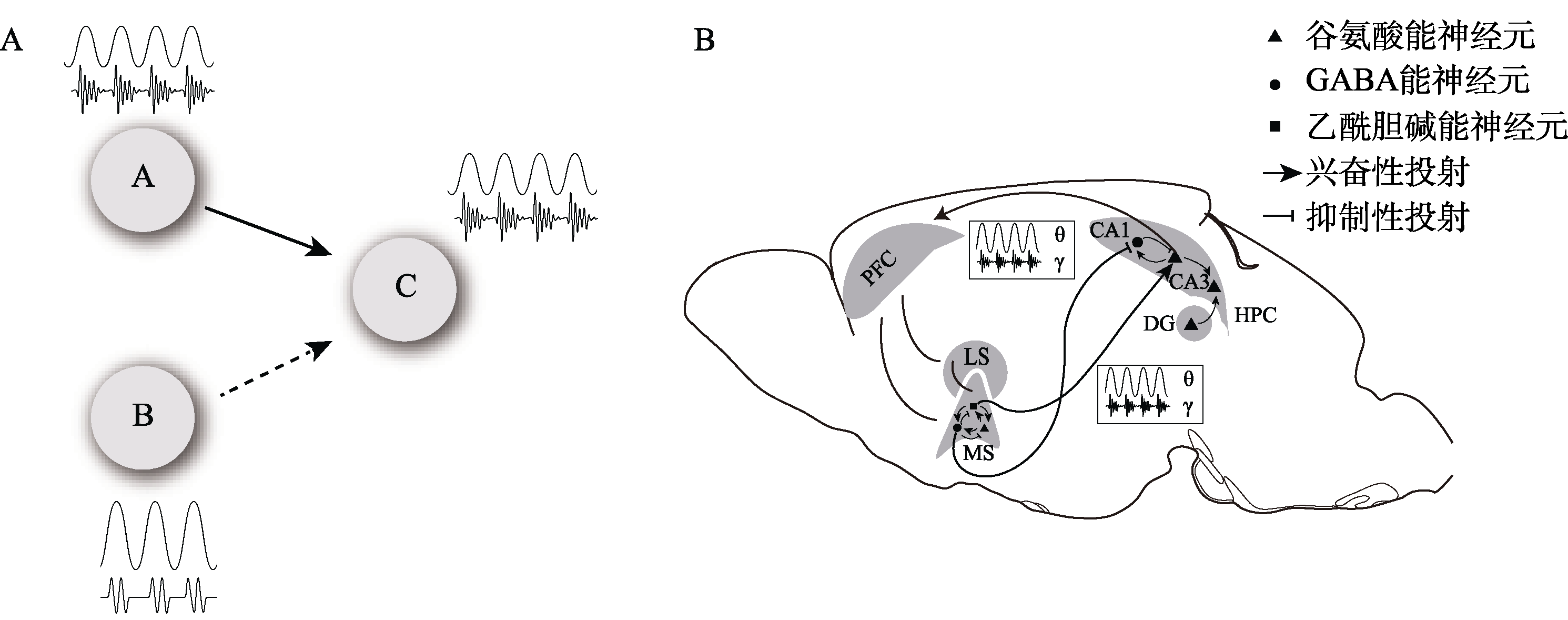
图3 低频振荡(如theta振荡)相位和gamma功率之间的耦合可以促进跨区域之间信息的同频协调。(A)脑区A和C中的神经元进行通信时, 它们通过低频(如theta振荡)波段进行连贯的同频振荡, 通常gamma振荡嵌套在theta振荡中, 即TG-PAC。因此脑区A中的gamma活动将能够传递到C区中的神经元, A和C中的gamma振荡将保持同频。在脑区B则作为脑区C低频振荡的外部重要驱动源, 间接影响脑区A和脑区C之间的TG-PAC。(B)图中三角形、圆形和正方形分别代表谷氨酸能神经元、GABA能神经元和胆碱能神经元, 图中显示了前额叶、内侧隔核和海马三者之间的神经联系, 其中内侧隔核内部三种神经元局部相互连接, 形成密集的局部网络, 内侧隔核的GABA能神经元和谷氨酸能神经元主要终止于海马CA1区的GABA能神经元; 海马CA1的谷氨酸能神经元投射至前额叶区域; 前额叶则与内侧隔核和外侧隔核均有突触联系。前额叶与海马、内侧隔核与海马之间存在的theta-gamma相位幅值耦合也以波形的方式在三个脑区之间标出。
| [1] | 张力新, 王发颀, 王玲, 杨佳佳, 万柏坤. (2017). 认知功能研究中神经振荡交叉节律耦合应用研究进展. 生理学报, 69(6), 805-816. https://doi.org/10.13294/j.aps.2017.0041 |
| [2] |
Abad-Perez, P., Molina-Payá, F. J., Martínez-Otero, L., Borrell, V., Redondo, R. L., & Brotons-Mas, J. R. (2023). Theta/ gamma co-modulation disruption after NMDAr blockade by MK-801 is associated with spatial working memory deficits in mice. Neuroscience, 519, 162-176. https://doi.org/10.1016/j.neuroscience.2023.03.022
doi: 10.1016/j.neuroscience.2023.03.022 URL |
| [3] |
Adams, E. J., Nguyen, A. T., & Cowan, N. (2018). Theories of working memory: Differences in definition, degree of modularity, role of attention, and purpose. Language, Speech, and Hearing Services in Schools, 49(3), 340-355. https://doi.org/10.1044/2018_LSHSS-17-0114
doi: 10.1044/2018_LSHSS-17-0114 URL |
| [4] |
Alekseichuk, I., Turi, Z., Amador de Lara, G., Antal, A., & Paulus, W. (2016). Spatial working memory in humans depends on theta and high gamma synchronization in the prefrontal cortex. Current Biology, 26(12), 1513-1521. https://doi.org/10.1016/j.cub.2016.04.035
doi: S0960-9822(16)30358-X URL pmid: 27238283 |
| [5] |
Axmacher, N., Henseler, M. M., Jensen, O., Weinreich, I., Elger, C. E., & Fell, J. (2010). Cross-frequency coupling supports multi-item working memory in the human hippocampus. Proceedings of the National Academy of Sciences of the United States of America, 107(7), 3228-3233. https://doi.org/10.1073/pnas.0911531107
doi: 10.1073/pnas.0911531107 URL pmid: 20133762 |
| [6] |
Baddeley, A. (2003). Working memory: Looking back and looking forward. Nature Reviews. Neuroscience, 4(10), 829-839. https://doi.org/10.1038/nrn1201
doi: 10.1038/nrn1201 URL pmid: 14523382 |
| [7] |
Barr, M. S., Rajji, T. K., Zomorrodi, R., Radhu, N., George, T. P., Blumberger, D. M., & Daskalakis, Z. J. (2017). Impaired theta-gamma coupling during working memory performance in schizophrenia. Schizophrenia Research, 189, 104-110. https://doi.org/10.1016/j.schres.2017.01.044
doi: S0920-9964(17)30057-9 URL pmid: 28148460 |
| [8] |
Benchenane, K., Peyrache, A., Khamassi, M., Tierney, P. L., Gioanni, Y., Battaglia, F. P., & Wiener, S. I. (2010). Coherent theta oscillations and reorganization of spike timing in the hippocampal- prefrontal network upon learning. Neuron, 66(6), 921-936. https://doi.org/10.1016/j.neuron.2010.05.013
doi: 10.1016/j.neuron.2010.05.013 URL pmid: 20620877 |
| [9] |
Benchenane, K., Tiesinga, P. H., & Battaglia, F. P. (2011). Oscillations in the prefrontal cortex: A gateway to memory and attention. Current Opinion in Neurobiology, 21(3), 475-485. https://doi.org/10.1016/j.conb.2011.01.004
doi: 10.1016/j.conb.2011.01.004 URL pmid: 21429736 |
| [10] | Bonnefond, M., Kastner, S., & Jensen, O. (2017). Communication between brain areas based on nested oscillations. eNeuro, 4(2), e0153-16.2017. https://doi.org/10.1523/ENEURO.0153-16.2017 |
| [11] |
Bortz, D. M., Feistritzer, C. M., & Grace, A. A. (2023). Medial prefrontal cortex to medial septum pathway activation improves cognitive flexibility in rats. The International Journal of Neuropsychopharmacology, 26(6), 426-437. https://doi.org/10.1093/ijnp/pyad019
doi: 10.1093/ijnp/pyad019 URL |
| [12] |
Buchanan, S. L., Thompson, R. H., Maxwell, B. L., & Powell, D. A. (1994). Efferent connections of the medial prefrontal cortex in the rabbit. Experimental Brain Research, 100(3), 469-483. https://doi.org/10.1007/BF02738406
doi: 10.1007/BF02738406 URL pmid: 7529194 |
| [13] |
Buzsáki, G. (2002). Theta oscillations in the hippocampus. Neuron, 33(3), 325-340. https://doi.org/10.1016/s0896-6273(02)00586-x
doi: 10.1016/s0896-6273(02)00586-x URL pmid: 11832222 |
| [14] | Cassaday, H. J., Nelson, A. J. D., & Pezze, M. A. (2014). From attention to memory along the dorsal-ventral axis of the medial prefrontal cortex: Some methodological considerations. Frontiers in Systems Neuroscience, 8, 160. https://doi.org/10.3389/fnsys.2014.00160 |
| [15] |
Chai, W. J., Abd Hamid, A. I., & Abdullah, J. M. (2018). Working memory from the psychological and neurosciences perspectives: A review. Frontiers in Psychology, 9, 401. https://doi.org/10.3389/fpsyg.2018.00401
doi: 10.3389/fpsyg.2018.00401 URL |
| [16] |
Chaieb, L., Leszczynski, M., Axmacher, N., Höhne, M., Elger, C., E., & Fell, J. (2015). Theta-gamma phase-phase coupling during working memory maintenance in the human hippocampus. Cognitive Neuroscience, 6(4), 149-157. https://doi.org/10.1080/17588928.2015.1058254
doi: 10.1080/17588928.2015.1058254 URL pmid: 26101947 |
| [17] |
Chiba, T., Kayahara, T., & Nakano, K. (2001). Efferent projections of infralimbic and prelimbic areas of the medial prefrontal cortex in the Japanese monkey, Macaca fuscata. Brain Research, 888(1), 83-101. https://doi.org/10.1016/s0006-8993(00)03013-4
doi: 10.1016/s0006-8993(00)03013-4 URL pmid: 11146055 |
| [18] |
Cissé, R. S., Krebs-Kraft, D. L., & Parent, M. B. (2008). Septal infusions of the hyperpolarization-activated cyclic nucleotide-gated channel (HCN-channel) blocker ZD7288 impair spontaneous alternation but not inhibitory avoidance. Behavioral Neuroscience, 122(3), 549-556. https://doi.org/10.1037/0735-7044.122.3.549
doi: 10.1037/0735-7044.122.3.549 URL pmid: 18513125 |
| [19] |
Colgin, L. L., Denninger, T., Fyhn, M., Hafting, T., Bonnevie, T., Jensen, O., Moser, M. -B., & Moser, E. I. (2009). Frequency of gamma oscillations routes flow of information in the hippocampus. Nature, 462(7271), 353-357. https://doi.org/10.1038/nature08573
doi: 10.1038/nature08573 URL |
| [20] |
Colgin, L. L., & Moser, E. I. (2010). Gamma oscillations in the hippocampus. Physiology, 25(5), 319-329. https://doi.org/10.1152/physiol.00021.2010
doi: 10.1152/physiol.00021.2010 URL pmid: 20940437 |
| [21] |
Constantinidis, C., & Goldman-Rakic, P. S. (2002). Correlated discharges among putative pyramidal neurons and interneurons in the primate prefrontal cortex. Journal of Neurophysiology, 88(6), 3487-3497. https://doi.org/10.1152/jn.00188.2002
URL pmid: 12466463 |
| [22] |
Curtis, C. E., & Sprague, T. C. (2021). Persistent activity during working memory from front to back. Frontiers in Neural Circuits, 15, 696060. https://doi.org/10.3389/fncir.2021.696060
doi: 10.3389/fncir.2021.696060 URL |
| [23] |
Daume, J., Kamiński, J., Schjetnan, A. G. P., Salimpour, Y., Khan, U., Kyzar, M., … Rutishauser, U. (2024). Control of working memory by phase-amplitude coupling of human hippocampal neurons. Nature, 629(8011), 393-401. https://doi.org/10.1038/s41586-024-07309-z
doi: 10.1038/s41586-024-07309-z URL |
| [24] |
De Almeida, L., Idiart, M., & Lisman, J. E. (2009). A second function of gamma frequency oscillations: An E%-max winner-take-all mechanism selects which cells fire. The Journal of Neuroscience, 29(23), 7497-7503. https://doi.org/10.1523/JNEUROSCI.6044-08.2009
doi: 10.1523/JNEUROSCI.6044-08.2009 URL |
| [25] |
De Mooij-van Malsen, J. G., Röhrdanz, N., Buschhoff, A. -S., Schiffelholz, T., Sigurdsson, T., & Wulff, P. (2023). Task- specific oscillatory synchronization of prefrontal cortex, nucleus reuniens, and hippocampus during working memory. iScience, 26(9), 107532. https://doi.org/10.1016/j.isci.2023.107532
doi: 10.1016/j.isci.2023.107532 URL |
| [26] |
Fernández, A., Pinal, D., Díaz, F., & Zurrón, M. (2021). Working memory load modulates oscillatory activity and the distribution of fast frequencies across frontal theta phase during working memory maintenance. Neurobiology of Learning and Memory, 183, 107476. https://doi.org/10.1016/j.nlm.2021.107476
doi: 10.1016/j.nlm.2021.107476 URL |
| [27] |
Fries, P. (2015). Rhythms for cognition: Communication through coherence. Neuron, 88(1), 220-235. https://doi.org/10.1016/j.neuron.2015.09.034
doi: 10.1016/j.neuron.2015.09.034 URL pmid: 26447583 |
| [28] |
Fuster, J. M., & Alexander, G. E. (1971). Neuron activity related to short-term memory. Science, 173(3997), 652-654. https://doi.org/10.1126/science.173.3997.652
doi: 10.1126/science.173.3997.652 URL pmid: 4998337 |
| [29] | Gemzik, Z. M., Donahue, M. M., & Griffin, A. L. (2021). Optogenetic suppression of the medial septum impairs working memory maintenance. Learning & Memory, 28(10), 361-370. https://doi.org/10.1101/lm.053348.120 |
| [30] | Gemzik, Z. M., & Griffin, A. L. (2025). Medial septal theta stimulation enhances spatial working memory performance in rats. Learning & Memory, 32(4), a054075. https://doi.org/10.1101/lm.054075.124 |
| [31] |
Gregoriou, G. G., Gotts, S. J., Zhou, H., & Desimone, R. (2009). High-frequency, long-range coupling between prefrontal and visual cortex during attention. Science, 324(5931), 1207-1210. https://doi.org/10.1126/science.1171402
doi: 10.1126/science.1171402 URL pmid: 19478185 |
| [32] | György, B. (2006). Rhythms of the brain (pp. 136-174). Oxford University Press. https://doi.org/10.1093/acprof:oso/9780195301069.001.0001 |
| [33] | He, Y., Guo, W., Ren, Z., Liu, S., & Ming, D. (2023). Gamma rhythm and theta-gamma coupling alternation in chronic unpredictable stress (CUS)-induced depression rats. 2023 45th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (pp.1-4). Sydney, Australia. https://doi.org/10.1109/EMBC40787.2023.10340209 |
| [34] |
Jensen, O., & Colgin, L. L. (2007). Cross-frequency coupling between neuronal oscillations. Trends in Cognitive Sciences, 11(7), 267-269. https://doi.org/10.1016/j.tics.2007.05.003
doi: 10.1016/j.tics.2007.05.003 URL pmid: 17548233 |
| [35] |
Jimura, K., Chushak, M. S., Westbrook, A., & Braver, T. S. (2018). Intertemporal decision-making involves prefrontal control mechanisms associated with working memory. Cerebral Cortex, 28(4), 1105-1116. https://doi.org/10.1093/cercor/bhx015
doi: 10.1093/cercor/bhx015 URL |
| [36] |
Jones, K. T., Johnson, E. L., & Berryhill, M. E. (2020). Frontoparietal theta-gamma interactions track working memory enhancement with training and tDCS. NeuroImage, 211, 116615. https://doi.org/10.1016/j.neuroimage.2020.116615
doi: 10.1016/j.neuroimage.2020.116615 URL |
| [37] |
Kim, C., Kroger, J. K., Calhoun, V. D., & Clark, V. P. (2015). The role of the frontopolar cortex in manipulation of integrated information in working memory. Neuroscience Letters, 595, 25-29. https://doi.org/10.1016/j.neulet.2015.03.044
doi: 10.1016/j.neulet.2015.03.044 URL pmid: 25818331 |
| [38] |
Király, B., Domonkos, A., Jelitai, M., Lopes-Dos-Santos, V., Martínez-Bellver, S., Kocsis, B., … Hangya, B. (2023). The medial septum controls hippocampal supra-theta oscillations. Nature Communications, 14(1), 6159. https://doi.org/10.1038/s41467-023-41746-0
doi: 10.1038/s41467-023-41746-0 URL |
| [39] |
Kramis, R., Vanderwolf, C. H., & Bland, B. H. (1975). Two types of hippocampal rhythmical slow activity in both the rabbit and the rat: Relations to behavior and effects of atropine, diethyl ether, urethane, and pentobarbital. Experimental Neurology, 49(1), 58-85. https://doi.org/10.1016/0014-4886(75)90195-8
doi: 10.1016/0014-4886(75)90195-8 URL |
| [40] |
Leung, L. S. (1998). Generation of theta and gamma rhythms in the hippocampus. Neuroscience and Biobehavioral Reviews, 22(2), 275-290. https://doi.org/10.1016/s0149-7634(97)00014-6
doi: 10.1016/s0149-7634(97)00014-6 URL pmid: 9579318 |
| [41] |
Leutgeb, S., Leutgeb, J. K., Barnes, C. A., Moser, E. I., McNaughton, B. L., & Moser, M. -B. (2005). Independent codes for spatial and episodic memory in hippocampal neuronal ensembles. Science, 309(5734), 619-623. https://doi.org/10.1126/science.1114037
URL pmid: 16040709 |
| [42] |
Li, J., Cao, D., Yu, S., Xiao, X., Imbach, L., Stieglitz, L., Sarnthein, J., & Jiang, T. (2023). Functional specialization and interaction in the amygdala-hippocampus circuit during working memory processing. Nature Communications, 14(1), 2921. https://doi.org/10.1038/s41467-023-38571-w
doi: 10.1038/s41467-023-38571-w URL |
| [43] |
Li, L. -B., Zhang, L., Sun, Y. -N., Han, L. -N., Wu, Z. -H., Zhang, Q. -J., & Liu, J. (2015). Activation of serotonin2A receptors in the medial septum-diagonal band of Broca complex enhanced working memory in the hemiparkinsonian rats. Neuropharmacology, 91, 23-33. https://doi.org/10.1016/j.neuropharm.2014.11.025
doi: 10.1016/j.neuropharm.2014.11.025 URL pmid: 25486618 |
| [44] |
Lisman, J., & Buzsáki, G. (2008). A neural coding scheme formed by the combined function of gamma and theta oscillations. Schizophrenia Bulletin, 34(5), 974-980. https://doi.org/10.1093/schbul/sbn060
doi: 10.1093/schbul/sbn060 URL pmid: 18559405 |
| [45] |
Lisman, J. E., & Idiart, M. A. (1995). Storage of 7 +/- 2 short-term memories in oscillatory subcycles. Science, 267(5203), 1512-1515. https://doi.org/10.1126/science.7878473
doi: 10.1126/science.7878473 URL pmid: 7878473 |
| [46] |
Lisman, J. E., & Jensen, O. (2013). The θ-γ neural code. Neuron, 77(6), 1002-1016. https://doi.org/10.1016/j.neuron.2013.03.007
doi: 10.1016/j.neuron.2013.03.007 URL pmid: 23522038 |
| [47] |
Liu, J., Tabisola, K. M., & Morilak, D. A. (2025). A projection from the medial prefrontal cortex to the lateral septum modulates coping behavior on the shock-probe test. Neuropsychopharmacology, 50(8), 1245-1255. https://doi.org/10.1038/s41386-025-02074-7
doi: 10.1038/s41386-025-02074-7 URL |
| [48] |
López-Vázquez, M. Á., López-Loeza, E., Lajud Ávila, N., Gutiérrez-Guzmán, B. E., Hernández-Pérez, J. J., Reyes, Y. E., & Olvera-Cortés, M. E. (2014). Septal serotonin depletion in rats facilitates working memory in the radial arm maze and increases hippocampal high-frequency theta activity. European Journal of Pharmacology, 734, 105-113. https://doi.org/10.1016/j.ejphar.2014.04.005
doi: 10.1016/j.ejphar.2014.04.005 URL pmid: 24742376 |
| [49] |
Manseau, F., Danik, M., & Williams, S. (2005). A functional glutamatergic neurone network in the medial septum and diagonal band area. The Journal of Physiology, 566(3), 865-884. https://doi.org/10.1113/jphysiol.2005.089664
doi: 10.1113/jphysiol.2005.089664 URL |
| [50] |
Moore, A. B., Li, Z., Tyner, C. E., Hu, X., & Crosson, B. (2013). Bilateral basal ganglia activity in verbal working memory. Brain and Language, 125(3), 316-323. https://doi.org/10.1016/j.bandl.2012.05.003
doi: 10.1016/j.bandl.2012.05.003 URL pmid: 22854261 |
| [51] |
Müller, C., & Remy, S. (2018). Septo-hippocampal interaction. Cell and Tissue Research, 373(3), 565-575. https://doi.org/10.1007/s00441-017-2745-2
doi: 10.1007/s00441-017-2745-2 URL pmid: 29250747 |
| [52] |
Murty, V. P., Sambataro, F., Radulescu, E., Altamura, M., Iudicello, J., Zoltick, B., … Mattay, V. S. (2011). Selective updating of working memory content modulates meso- cortico-striatal activity. NeuroImage, 57(3), 1264-1272. https://doi.org/10.1016/j.neuroimage.2011.05.006
doi: 10.1016/j.neuroimage.2011.05.006 URL |
| [53] |
O’Keefe, J., & Conway, D. H. (1978). Hippocampal place units in the freely moving rat: Why they fire where they fire. Experimental Brain Research, 31(4), 573-590. https://doi.org/10.1007/BF00239813
doi: 10.1007/BF00239813 URL pmid: 658182 |
| [54] |
O’Neill, P. -K., Gordon, J. A., & Sigurdsson, T. (2013). Theta oscillations in the medial prefrontal cortex are modulated by spatial working memory and synchronize with the hippocampus through its ventral subregion. The Journal of Neuroscience, 33(35), 14211-14224. https://doi.org/10.1523/JNEUROSCI.2378-13.2013
doi: 10.1523/JNEUROSCI.2378-13.2013 URL |
| [55] |
Pouille, F., & Scanziani, M. (2001). Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science, 293(5532), 1159-1163. https://doi.org/10.1126/science.1060342
doi: 10.1126/science.1060342 URL pmid: 11498596 |
| [56] |
Roland, J. J., Stewart, A. L., Janke, K. L., Gielow, M. R., Kostek, J. A., Savage, L. M., Servatius, R. J., & Pang,K. C. H. (2014). Medial septum-diagonal band of Broca (MSDB) GABAergic regulation of hippocampal acetylcholine efflux is dependent on cognitive demands. The Journal of Neuroscience, 34(2), 506-514. https://doi.org/10.1523/JNEUROSCI.2352-13.2014
doi: 10.1523/JNEUROSCI.2352-13.2014 URL |
| [57] |
Rose, N. S., LaRocque, J. J., Riggall, A. C., Gosseries, O., Starrett, M. J., Meyering, E. E., & Postle, B. R. (2016). Reactivation of latent working memories with transcranial magnetic stimulation. Science, 354(6316), 1136-1139. https://doi.org/10.1126/science.aah7011
URL pmid: 27934762 |
| [58] |
Schneider, M., Walter, H., Moessnang, C., Schäfer, A., Erk, S., Mohnke, S., … Tost, H. (2017). Altered DLPFC- hippocampus connectivity during working memory: Independent replication and disorder specificity of a putative genetic risk phenotype for schizophrenia. Schizophrenia Bulletin, 43(5), 1114-1122. https://doi.org/10.1093/schbul/sbx001
doi: 10.1093/schbul/sbx001 URL pmid: 28207073 |
| [59] |
Sesack, S. R., Deutch, A. Y., Roth, R. H., & Bunney, B. S. (1989). Topographical organization of the efferent projections of the medial prefrontal cortex in the rat: An anterograde tract-tracing study with Phaseolus vulgaris leucoagglutinin. The Journal of Comparative Neurology, 290(2), 213-242. https://doi.org/10.1002/cne.902900205
doi: 10.1002/cne.v290:2 URL |
| [60] |
Shirvalkar, P. R., Rapp, P. R., & Shapiro, M. L. (2010). Bidirectional changes to hippocampal theta-gamma comodulation predict memory for recent spatial episodes. Proceedings of the National Academy of Sciences of the United States of America, 107(15), 7054-7059. https://doi.org/10.1073/pnas.0911184107
doi: 10.1073/pnas.0911184107 URL pmid: 20351262 |
| [61] |
Singer, W. (1993). Synchronization of cortical activity and its putative role in information processing and learning. Annual Review of Physiology, 55, 349-374. https://doi.org/10.1146/annurev.ph.55.030193.002025
URL pmid: 8466179 |
| [62] |
Solari, N., & Hangya, B. (2018). Cholinergic modulation of spatial learning, memory and navigation. The European Journal of Neuroscience, 48(5), 2199-2230. https://doi.org/10.1111/ejn.14089
doi: 10.1111/ejn.2018.48.issue-5 URL |
| [63] |
Spellman, T., Rigotti, M., Ahmari, S. E., Fusi, S., Gogos, J. A., & Gordon, J. A. (2015). Hippocampal-prefrontal input supports spatial encoding in working memory. Nature, 522(7556), 309-314. https://doi.org/10.1038/nature14445
doi: 10.1038/nature14445 URL |
| [64] |
Stackman, R. W., & Walsh, T. J. (1992). Chlordiazepoxide- induced working memory impairments: Site specificity and reversal by flumazenil (RO15-1788). Behavioral and Neural Biology, 57(3), 233-243. https://doi.org/10.1016/0163-1047(92)90206-j
URL pmid: 1319705 |
| [65] |
Swanson, L. W., & Cowan, W. M. (1979). The connections of the septal region in the rat. The Journal of Comparative Neurology, 186(4), 621-655. https://doi.org/10.1002/cne.901860408
doi: 10.1002/cne.v186:4 URL |
| [66] |
Takeuchi, Y., Nagy, A. J., Barcsai, L., Li, Q., Ohsawa, M., Mizuseki, K., & Berényi, A. (2021). The medial septum as a potential target for treating brain disorders associated with oscillopathies. Frontiers in Neural Circuits, 15, 701080. https://doi.org/10.3389/fncir.2021.701080
doi: 10.3389/fncir.2021.701080 URL |
| [67] | Tang, Y., Xing, Y., Sun, L., Wang, Z., Wang, C., Yang, K., … Zhao, G. (2024). Transcranial alternating current stimulation for patients with mild Alzheimer’s disease (TRANSFORM- AD): A randomized controlled clinical trial. Alzheimer’s Research & Therapy, 16(1), 203. https://doi.org/10.1186/s13195-024-01570-0 |
| [68] |
Tort, A. B. L., Kramer, M. A., Thorn, C., Gibson, D. J., Kubota, Y., Graybiel, A. M., & Kopell, N. J. (2008). Dynamic cross-frequency couplings of local field potential oscillations in rat striatum and hippocampus during performance of a T-maze task. Proceedings of the National Academy of Sciences of the United States of America, 105(51), 20517-20522. https://doi.org/10.1073/pnas.0810524105
doi: 10.1073/pnas.0810524105 URL pmid: 19074268 |
| [69] |
Tsanov, M. (2018). Differential and complementary roles of medial and lateral septum in the orchestration of limbic oscillations and signal integration. The European Journal of Neuroscience, 48(8), 2783-2794. https://doi.org/10.1111/ejn.13746
doi: 10.1111/ejn.2018.48.issue-8 URL |
| [70] |
Unal, G., Joshi, A., Viney, T. J., Kis, V., & Somogyi, P. (2015). Synaptic targets of medial septal projections in the hippocampus and extrahippocampal cortices of the mouse. The Journal of Neuroscience, 35(48), 15812-15826. https://doi.org/10.1523/JNEUROSCI.2639-15.2015
doi: 10.1523/JNEUROSCI.2639-15.2015 URL |
| [71] |
Van Den Berg, M., Toen, D., Verhoye, M., & Keliris, G. A. (2023). Alterations in theta-gamma coupling and sharp wave-ripple, signs of prodromal hippocampal network impairment in the TgF344-AD rat model. Frontiers in Aging Neuroscience, 15, 1081058. https://doi.org/10.3389/fnagi.2023.1081058
doi: 10.3389/fnagi.2023.1081058 URL |
| [72] |
Von Der Malsburg, C. (1985). Nervous structures with dynamical links. Berichte Der Bunsengesellschaft Für Physikalische Chemie, 89(6), 703-710. https://doi.org/10.1002/bbpc.19850890625
doi: 10.1002/bbpc.v89:6 URL |
| [73] | Von Der Malsburg, C. (1994). The correlation theory of brain function. In E. Domany, J. L. van Hemmen, & K. Schulten (Eds), Models of Neural Networks: Temporal Aspects of Coding and Information Processing in Biological Systems (pp. 95-119). Springer. https://doi.org/10.1007/978-1-4612-4320-5_2 |
| [74] |
Wulff, P., Ponomarenko, A. A., Bartos, M., Korotkova, T. M., Fuchs, E. C., Bähner, F., … Monyer, H. (2009). Hippocampal theta rhythm and its coupling with gamma oscillations require fast inhibition onto parvalbumin-positive interneurons. Proceedings of the National Academy of Sciences of the United States of America, 106(9), 3561-3566. https://doi.org/10.1073/pnas.0813176106
doi: 10.1073/pnas.0813176106 URL pmid: 19204281 |
| [75] | Zhang, W., Guo, L., & Liu, D. (2022). Concurrent interactions between prefrontal cortex and hippocampus during a spatial working memory task. Brain Structure & Function, 227(5), 1735-1755. https://doi.org/10.1007/s00429-022-02469-y |
| [76] |
Zhang, X., Zhong, W., Brankačk, J., Weyer, S. W., Müller, U. C., Tort, A. B. L., & Draguhn, A. (2016). Impaired theta- gamma coupling in APP-deficient mice. Scientific Reports, 6, 21948. https://doi.org/10.1038/srep21948
doi: 10.1038/srep21948 URL |
| [77] | Zhang, Y., Cao, L., Varga, V., Jing, M., Karadas, M., Li, Y., & Buzsáki, G. (2021). Cholinergic suppression of hippocampal sharp-wave ripples impairs working memory. Proceedings of the National Academy of Sciences of the United States of America, 118(15), e2016432118. https://doi.org/10.1073/pnas.2016432118 |
| [78] |
Zhang, Y., Zhang, Y., Yu, H., Yang, Y., Li, W., & Qian, Z. (2017). Theta-gamma coupling in hippocampus during working memory deficits induced by low frequency electromagnetic field exposure. Physiology & Behavior, 179, 135-142. https://doi.org/10.1016/j.physbeh.2017.05.033
doi: 10.1016/j.physbeh.2017.05.033 URL |
| [79] |
Zutshi, I., Brandon, M. P., Fu, M. L., Donegan, M. L., Leutgeb, J. K., & Leutgeb, S. (2018). Hippocampal neural circuits respond to optogenetic pacing of theta frequencies by generating accelerated oscillation frequencies. Current Biology, 28(8), 1179-1188.e3. https://doi.org/10.1016/j.cub.2018.02.061
doi: S0960-9822(18)30246-X URL pmid: 29628373 |
| [1] | 田仁霞, 杨平, 郭园园, 吴瑕. 治疗孤独症谱系障碍:重复经颅磁刺激的潜在作用[J]. 心理科学进展, 2025, 33(4): 598-610. |
| [2] | 吴际, 李会杰. 认知空间映射及其神经机制[J]. 心理科学进展, 2025, 33(1): 62-76. |
| [3] | 冯攀, 赵恒越, 姜雨矇, 张悦彤, 冯廷勇. 催产素影响条件化恐惧情绪加工的认知机制及神经基础[J]. 心理科学进展, 2024, 32(4): 557-567. |
| [4] | 周帆, 田昊月, 姜英杰. 记忆快速巩固:基于图式的学习与重复再激活[J]. 心理科学进展, 2024, 32(11): 1854-1871. |
| [5] | 周士人, 仇秀芙, 何振宏, 张丹丹. 基于无损脑刺激的情绪调节干预[J]. 心理科学进展, 2023, 31(8): 1477-1495. |
| [6] | 王雪珂, 冯廷勇. “冷”/“热”执行功能缺陷影响ADHD儿童核心症状的作用机制[J]. 心理科学进展, 2023, 31(11): 2106-2128. |
| [7] | 郭志华, 卢宏亮, 黄鹏, 朱霞. 经颅直流电刺激对健康人群反应抑制的影响[J]. 心理科学进展, 2022, 30(9): 2034-2052. |
| [8] | 冯攀, 杨可, 冯廷勇. 催产素影响恐惧习得和消退的认知神经机制[J]. 心理科学进展, 2022, 30(2): 365-374. |
| [9] | 严万森, 刘苏姣, 张冉冉, 徐鹏. 强迫性特征在药物成瘾行为中的易感性及其前额叶-反奖赏系统神经基础[J]. 心理科学进展, 2021, 29(8): 1345-1357. |
| [10] | 叶超群, 林郁泓, 刘春雷. 创造力产生过程中的神经振荡机制[J]. 心理科学进展, 2021, 29(4): 697-706. |
| [11] | 薛冰, 王雪娇, 马宁, 高军. 催产素调控心理韧性:基于对海马的作用机制[J]. 心理科学进展, 2021, 29(2): 311-322. |
| [12] | 李婉如, 库逸轩. 急性应激影响工作记忆的生理心理机制[J]. 心理科学进展, 2020, 28(9): 1508-1524. |
| [13] | 张家鑫, 海拉干, 李会杰. 空间导航的测量及其在认知老化中的应用[J]. 心理科学进展, 2019, 27(12): 2019-2033. |
| [14] | 张慧会, 张亮. 早期应激对情绪调节的影响及其神经机制[J]. 心理科学进展, 2018, 26(7): 1193-1203. |
| [15] | 成梅, 杨燕, 尹华站. 经颅直流电刺激在注意缺陷多动障碍治疗中的应用[J]. 心理科学进展, 2018, 26(4): 657-666. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
